Published on Angewandte!
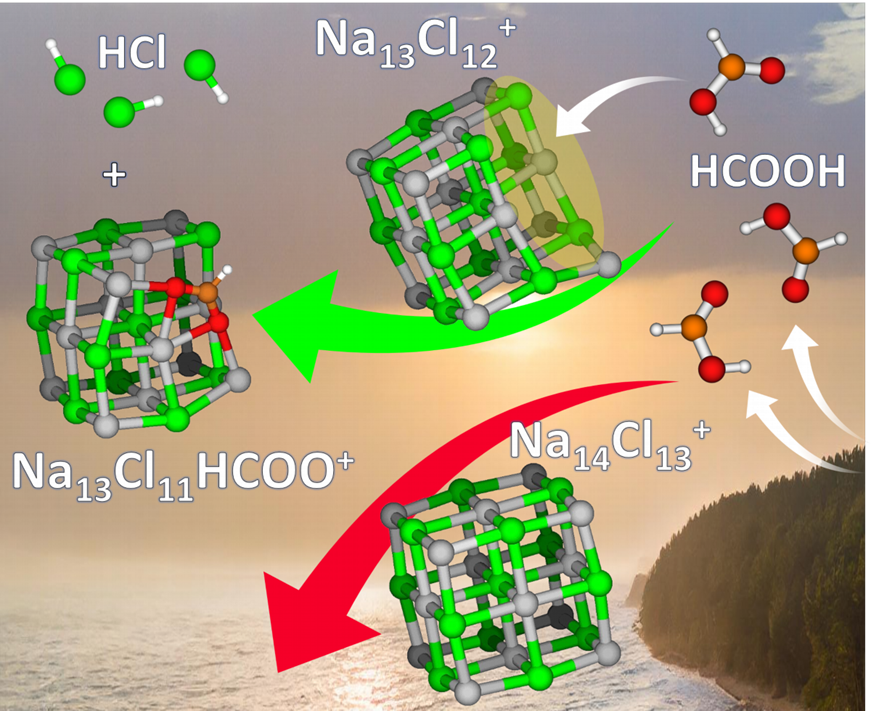
The acid displacement reaction of formic acid in sea-salt aerosols to release hydrogen chloride is an enigma in atmospheric chemistry: a weak acid transfers a proton to the conjugate base of a strong acid, reversing solution-phase acid–base chemistry. We show that a surface defect of the crystal lattice Na13Cl12+ reacts readily with release of hydrogen chloride and incorporation of formate into the salt structure. No reaction is observed with Na14Cl13+, a known magic cluster that corresponds to a cubic section of the crystal lattice. The reason lies in the induced fit of the formate ion in the Na13Cl11(HCOO)+ cluster. Its oxygen atoms interact with five sodium ions.
